Archived
Please note, this page may contain outdated information or subject matter.
 Researchers funded by Action are investigating whether a commonly used diabetes medicine could help babies who’ve suffered life-threatening brain damage at or around birth.
Researchers funded by Action are investigating whether a commonly used diabetes medicine could help babies who’ve suffered life-threatening brain damage at or around birth.
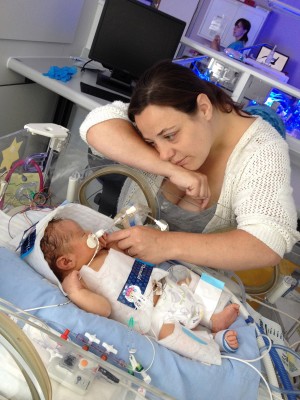
Aiden, pictured right, endured a traumatic start to life and his survival was uncertain. Complications at his birth meant he was deprived of oxygen for more than 20 minutes and needed prolonged resuscitation.
“For the first two hours I didn’t know whether our beautiful son had survived or not,” recalls his dad, Keith.
Doctors began to reduce Aiden’s body temperature to protect him from further brain damage, a process known as cooling therapy. This breakthrough treatment is now routine in the UK and was the product of a 20-year programme of research to which Action Medical Research contributed more than £1m.
“Aiden was cooled for 72 hours,” says Keith. “His little body was wrapped in an ice jacket helping prevent the spread of further brain damage. Without this vital treatment our little boy may not be here now.”
But while cooling therapy gives babies like Aiden a better chance of surviving and can protect against or at least limit disability, sadly it doesn’t save all babies. Neonatal encephalopathy (NE), a form of brain damage that is usually caused by a shortage of oxygen at birth, still kills around one in five affected babies in the UK. Many others develop lifelong problems such as cerebral palsy and learning disabilities.
Each year around 700,000 babies worldwide die or face lifelong disability due to brain damage at birth
Action has awarded funding of more than £193,000 to Dr Ahad Rahim of University College London, who says there is an overwhelming need for further improvements in the treatment of this condition.
Dr Rahim and his team are investigating whether a diabetes medicine, called exendin-4, could help more babies. The drug is thought to have protective effects on the brain and clinical trials are already underway to find out if it benefits people with Alzheimer’s and Parkinson’s diseases.
If successful, this means that it should be faster to develop it as a treatment for babies affected by NE rather than a totally new medicine. The drug could then be used either alone or in combination with cooling therapy to help more babies who are at risk.
Aiden’s mum, Fleur, says this new research is very important.
“Without previous research Aiden’s outcome could have been even more severe. By undergoing cooling we were given hope. NE affects increasing amounts of babies and those babies and their families deserve to have the hope that we did.”
